A Tragedy That Shook a Nation
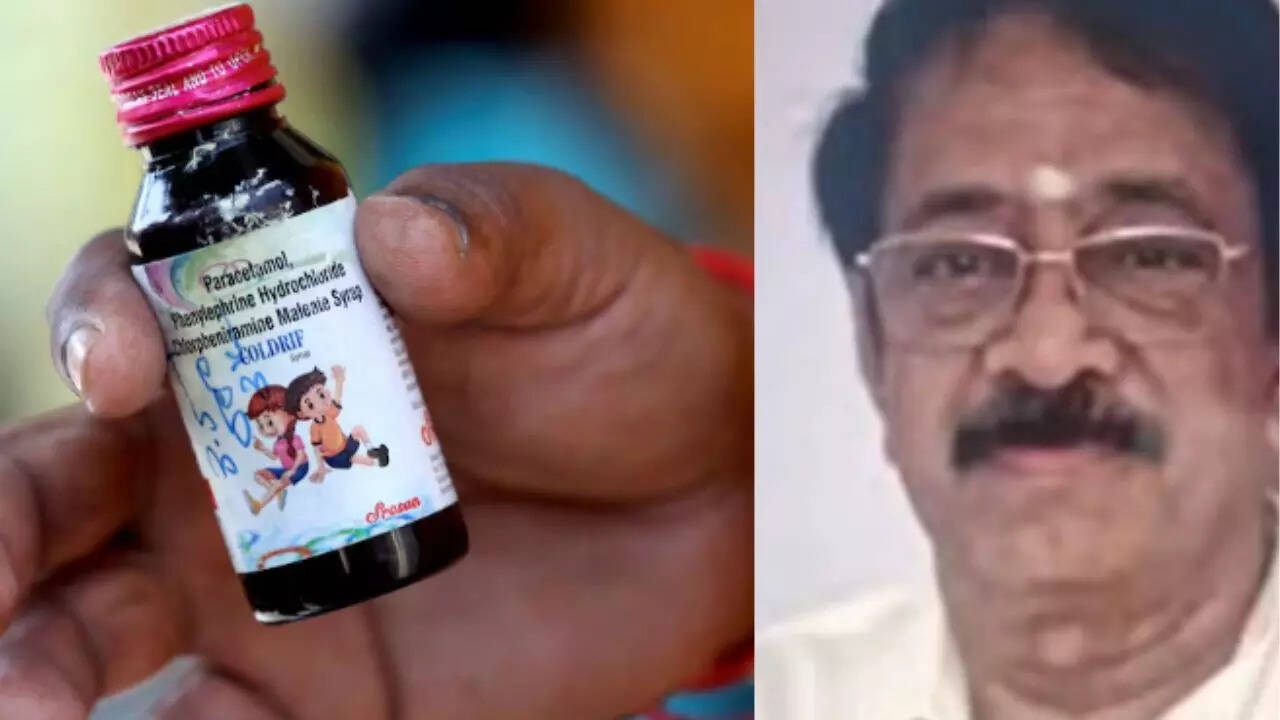
In a horrifying turn of events, what began as a routine prescription for children suffering from seasonal coughs and colds turned into a national nightmare. The “Coldrif” cough syrup, manufactured by Sresan Pharmaceuticals, was linked to the deaths of at least 20 children in Chhindwara, Madhya Pradesh, sending shockwaves across the country. Investigations revealed that the medicine contained dangerously high levels of toxic chemicals, sparking outrage, grief, and a massive hunt for those responsible.
After weeks of evasion, Govindan Ranganathan, the 75-year-old owner of Sresan Pharma, was arrested in a dramatic midnight operation in Chennai, bringing one chapter of this tragic saga to a close—but leaving deep questions about pharmaceutical oversight and accountability in India.
The Man Behind the Mask: Who Is Govindan Ranganathan?
Once regarded as a respected entrepreneur in Tamil Nadu’s pharmaceutical circles, Govindan Ranganathan built his career over four decades. A pharmacy graduate from Madras Medical College, he first gained attention in the 1980s with a nutritional supplement named Pronit, which he marketed personally to doctors. Though the product initially faced scrutiny over its ingredients, Ranganathan managed to secure regulatory clearances and continued expanding his business.
He eventually established Sresan Pharmaceuticals, operating out of a modest 2,000 sq ft unit on the Chennai–Bengaluru highway. The company also maintained ties with other small firms like Ceego Labs and Iven Healthcare, positioning itself as a producer of affordable syrups and tonics. But behind the image of a self-made businessman mentoring young pharmacists, investigators now say Ranganathan’s company was steeped in regulatory violations, unsafe practices, and negligence that ultimately cost innocent lives.
A Company Built on Neglect and Deceit
Sresan Pharmaceuticals’ story is one of long-term disregard for safety standards. Official records show that while the firm was registered as a private limited company in 1990, it was later struck off the Ministry of Corporate Affairs register, yet continued operations illegally under a proprietary model. The last official balance sheet dates back to 2009—a glaring sign of systemic oversight failure.
Investigations revealed that industrial-grade chemicals, unsuitable for human consumption, were used to make the Coldrif syrup. Key ingredients, including propylene glycol, were sourced from local traders and even paint industry suppliers, with payments made through cash and digital transfers to evade detection.
Testing confirmed the presence of 48.6% Diethylene Glycol (DEG)—a highly toxic compound known to cause acute kidney failure, neurological damage, and death. Officials discovered over 350 violations in the manufacturing process, including unhygienic production conditions. In response, several states including Madhya Pradesh, Rajasthan, Tamil Nadu, Kerala, and Uttar Pradesh immediately banned the sale of the syrup, while Karnataka and Telangana issued health alerts urging citizens to avoid it.
The Manhunt and Arrest
Following the tragic deaths, Ranganathan and his wife vanished, prompting a nationwide search. The Madhya Pradesh Police announced a ₹20,000 reward for any information on their whereabouts. Acting on specific intelligence, a special team comprising cyber experts, female officers, and drug inspectors tracked the fugitive to Chennai.
After days of surveillance—monitoring bank transactions, vehicle movements, and communication logs—the operation culminated at 1:30 a.m. on October 5, when Ranganathan was captured. He was taken to Sresan Pharma’s Kancheepuram facility, where crucial documents were seized, and the factory sealed. The police are now seeking transit remand to transfer him to Chhindwara, the epicenter of the tragedy.
Authorities have charged Ranganathan with culpable homicide not amounting to murder, adulteration of drugs, and endangering children’s lives. The investigation is expanding to examine chemical suppliers, distributors, and intermediaries involved in the tainted product’s supply chain.
Public Outrage and Government Response
The revelations have triggered widespread public anger and calls for stricter oversight of India’s pharmaceutical industry. Parents of the victims have demanded swift justice, while activists are urging reforms to close regulatory loopholes that allow unlicensed or dormant companies to operate unchecked.
The Union Health Ministry has ordered a comprehensive review of small-scale drug manufacturing units across India. A special task force has been formed to inspect compliance records, raw material sourcing, and testing protocols in pharmaceutical clusters, particularly in Tamil Nadu and Madhya Pradesh.
A Cautionary Tale of Profit Over Principles
The Coldrif tragedy is a grim reminder of what happens when greed eclipses ethics and oversight fails to protect the vulnerable. Twenty young lives were lost—not to disease, but to negligence masquerading as enterprise. The arrest of Govindan Ranganathan may bring legal closure, but true justice lies in systemic reform—ensuring that every bottle of medicine in India is as safe as it claims to be.
As the nation mourns, this tragedy must serve as a turning point—a moment to rebuild trust in an industry that sustains life, not one that endangers it.
(With agency inputs)